Treatment
Treatment options
Treatment for HE aims to normalise neurophysiological functions by eliminating causative factors and reducing ammonia levels. If successful, treatment results in improvement of cerebral neurotransmission, astroglial function and ultimately in clinical improvement. As the underlying disease is not eliminated the patient remains at risk of a reoccurrence of HE, even after successful therapy.1
Treatment should be initiated in the early stages of HE as patients suffer from fatigue, show disturbances of their sleeping and waking cycle and are at particular risk when driving and performing manual work.
Treatment should be given to any patient experiencing any form of clinically manifest HE or in a situation which could lead to clinically manifest HE. In subclinical HE, treatment should be given in accordance with the everyday demands on the patient i.e. their job and fitness to drive.1
The therapeutic possibilities in treating HE:1
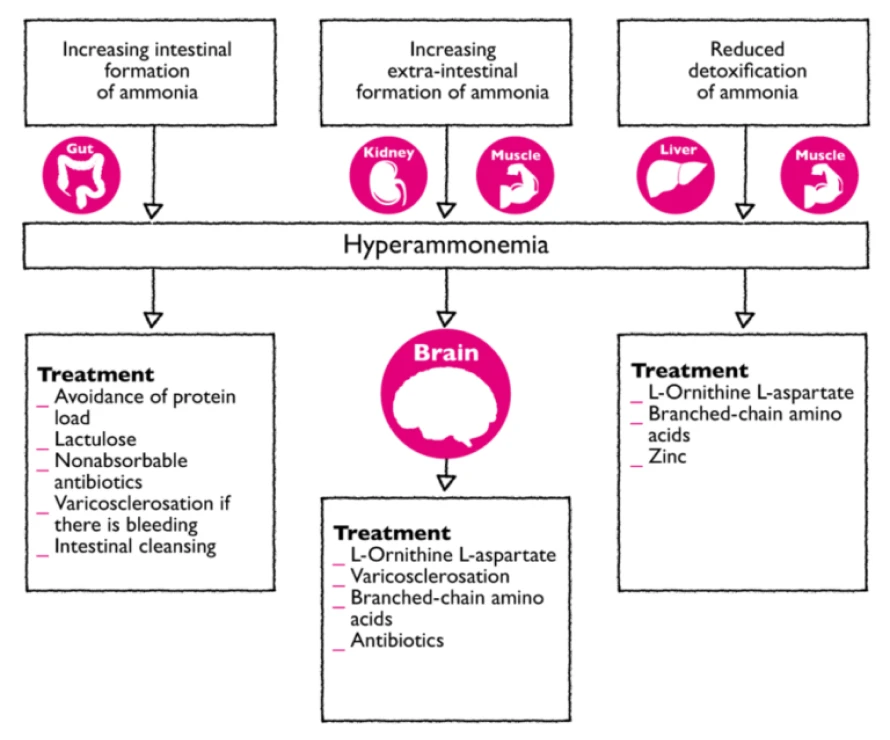
- Antibiotics1
The use of poorly absorbed antibiotics such (e.g., rifaximin, paromomycin, neomycin, metronidazole) has been indicated. Recommended dose is up to 4-6 g/d for at most 7 days. Rifaximin is now available for long-term use. These antibiotics act to reduce the physiological flora in the large intestine, therefore reducing the production of ammonia. This treatment option is expensive and associated with considerable side effects (diarrhoea, malabsorption, urticaria, infection ) with the added risk of antibiotic resistance.
Eliminate precipitating factors
The first step should be the identification and elimination of precipitating factors, as these can play a role in 70-80% of HE patients:1
- Stop gastrointestinal bleeding
- Treatment of anaemia (aim: haematocrit = 30%)
- Treatment of acidosis with bicarbonate
- Correction of electrolytes to within normal range
- Discontinue diuretics and sedatives
- Treatment of infections with antibiotics
- Abstinence from alcohol
Successfully eliminating the triggering factors can positively influence the progression of HE.
Decreasing ammonia production
Intestinal cleansing or inhibition of intestinal ammonia production would result in a decrease of ammoniagenic substrates. This can be accomplished by synthetic, non-absorbable disaccharides (lactulose) or non-absorbable antibiotics (e.g. rifaximin).1
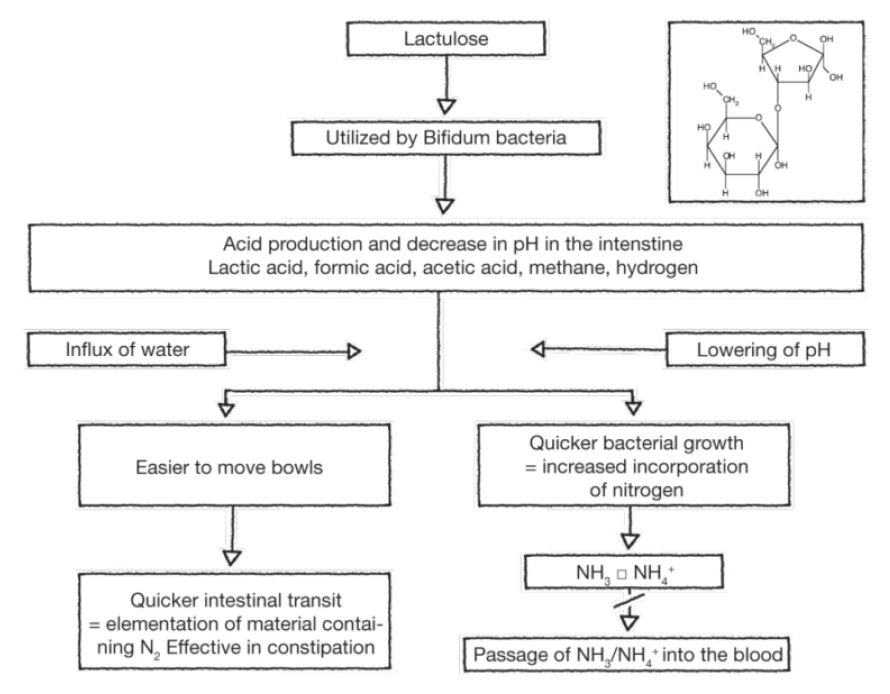
- Lactulose
Lactulose (along with lactilol) is a nonabsorbable disaccharide that has been in common clinical use since the early 1970s. Lactulose is degraded by intestinal bacteria to lactic acid and other organic acids. It appears to inhibit intestinal ammonia production by a number of mechanisms. The conversion of lactulose to lactic acid results in acidification of the gut lumen and this promotes conversion of NH4+ to NH3 and the subsequent passage of NH3 from tissues into the lumen. Gut acidification inhibits ammoniagenic coliform bacteria, leading to increased levels of nonammoniagenic lactobacilli. Lactulose also works as a cathartic, reducing the overall colonic bacterial load. A 2013 meta-analysis affirmed the clinical utility of lactulose in the management of HE.2
The mode of action of lactulose in the treatment of HE1
Increasing detoxification of ammonia in liver and muscle
- L-ornithine L-aspartate (LOLA; Hepa-Merz®)1
Therapeutic administration of LOLA increases ammonia affects detoxification in two ways:
- Activation of the urea cycle in the residual hepatocytes of the liver, through provision of the metabolic substrates ornithine and aspartate, these promote glutamine formation, thereby stimulating ammonia detoxification via glutamine synthesis in the liver, in the brain and muscle;
- In the presence of hyperammonemia and collateral circulation or a hepatic detoxification deficit, ammonia detoxification via glutamine synthesis in the brain and muscle tissue makes an additional contribution to the ammonia-lowering effect of LOLA.
- Benzodiazepine antagonists1
These work by inhibiting endogenous benzodiazepine excess thus counteracting the neurodepressive effects of GABAergic activation. Flumazenil (Anexate®) is a commonly used benzodiazepine antagonist.
The use of benzodiazepine antagonists in the treatment of HE has been supported by study data (> 90% improvement in HE III/IV)* and they are considered easy to use (0.5 mg i.v.). The principle disadvantages of this treatment option is that the efficacy is extremely short-lived (30-60 min, max in 1 study 5.5 h), also the effect is unpredictable in patients who have received prior treatment with exogenous benzodiazepines (e.g. for gastroscopy).
- Silymarin1
Silymarin is a phytopharmaceutical agent (extract of milk thistle seeds). The extract contains an active compound called flavonoid silibinin. It acts to protect the liver by
- Preventing entry of toxins into liver
- Supporting protein synthesis
- Stimulating regeneration of damaged hepatocytes
In clinical studies there is an improvement in laboratory parameters and histological findings (less fibrosis).
Summary of evidence-based effectiveness of drug therapy for treating HE
Therapeutic principle | Active substance | Results of placebo controlled trials |
Reduction of intestinal ammonia production | Lactulose enema | Better than placebo1 |
Oral lactulose | Better than placebo | |
Neomycin | Not better than placebo | |
Increading extra-intestinal ammonia detoxification | L-ornithine-L-aspartate | Better than placebo |
Correcting amino acid imbalance | LBrnached-chain amino acids (BCAA) | Better than placebo |
Benzodiacepine Rezeptor antagonist | Flumazenil | Better than placebo |