Hepa-Merz®
Mode of action
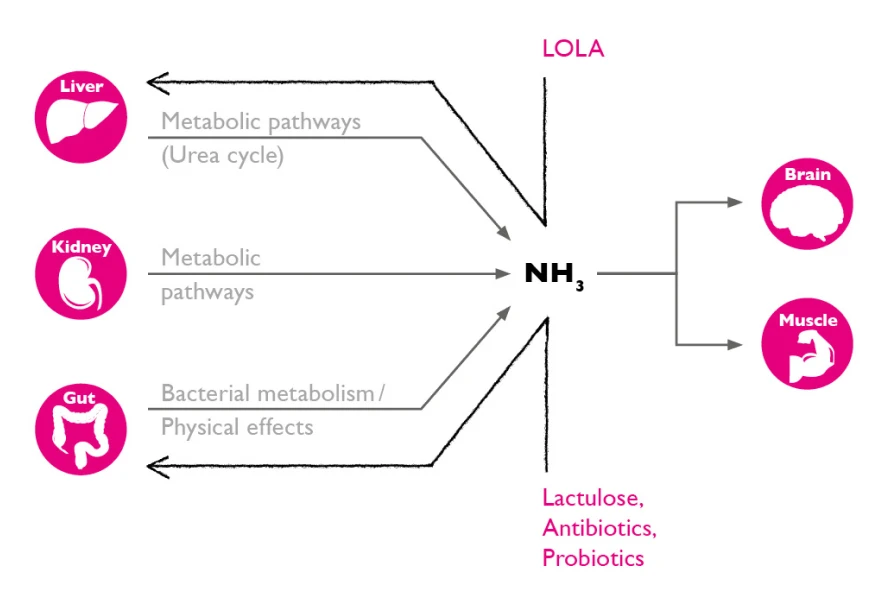
Hepa-Merz® targets systemic neurotoxic ammonia. By reducing serum ammonia concentration, LOLA benefits patients with both overt and minimal hepatic encephalopathy (HE).1-3
Bridging the gap between liver and brain: LOLA mechanism of action in treating HE
- 15% of ammonia production originates in the gut4
- The main contribution is from the liver
- LOLA interacts with metabolic pathways (urea cycle) supporting natural ammonia detoxification and maintaining natural balance in the gut1-4
Hepa-Merz® provides unique ammonia detoxification simultaneously in the liver and muscle via two distinct pathways:2,3
via glutamine synthesis
- in the brain
- in the muscle
- in the liver (perivenous cells)
via activation of the urea cycle
- in the liver (periportal hepatocytes)
These mechanisms support natural ammonia detoxification, maintaining the body’s natural balance in the gut. Importantly, systemic ammonia detoxification leads to reduction of HE symptoms and brain protection.1-4
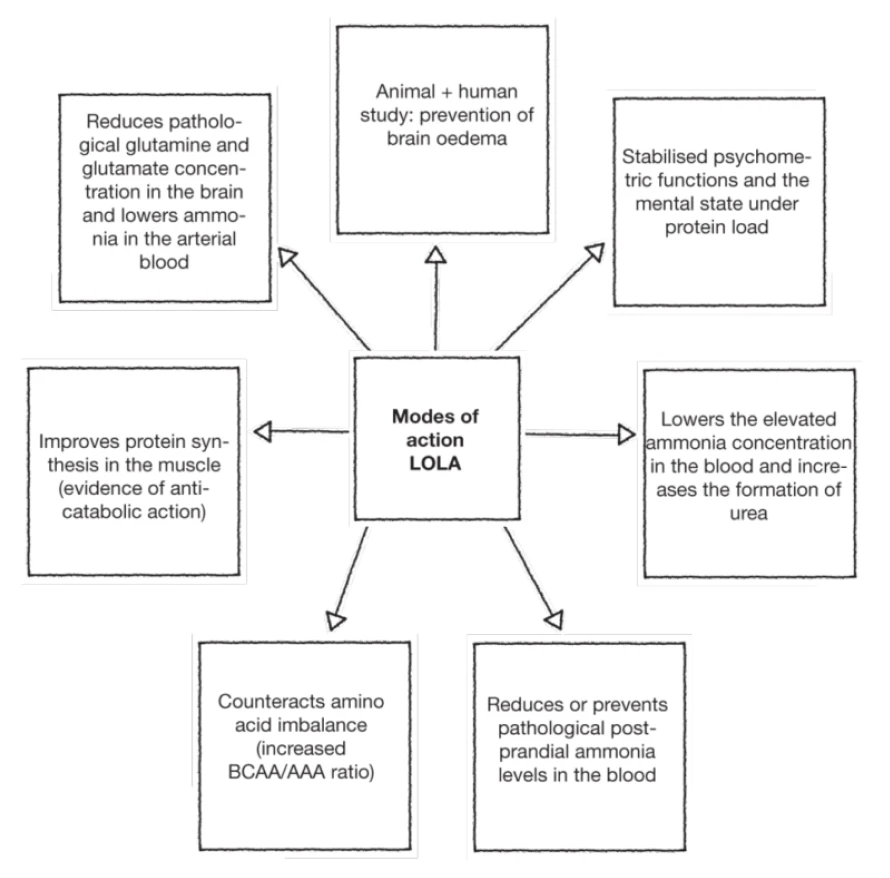
LOLA enhances ammonia detoxification by increasing urea synthesis. Urea synthesis is an irreversible and liver-specific process, the ornithine in LOLA is taken up by periportal hepatocytes.
- Ornithine acts as a substrate of urea cycle and also as an allosteric activator of the carbamoyl phosphate synthetase
- Aspartate acts as a substrate in the urea cycle
LOLA increases the reversible detoxification of ammonia systemically via glutamine synthesis in the brain and muscles. Aspartate and glutamate are taken up almost exclusively by perivenous liver cells. They serve as a carbon skeleton source to enhance the impaired glutamine synthesis in defective perivenous livers cells of cirrhotic patients.
- Ornithine is converted to glutamate as well and acts as a substrate in glutamine synthesis
- Aspartate cycle-derived glutamate acts as a substrate in glutamine synthesis
Overview of Hepa-Merz® mode of action1-4
1. Bai M et al. l-ornithine-l-aspartate for hepatic encephalopathy in patients with cirrhosis: a meta-analysis of randomized controlled trials. J Gastroenterol Hepatol. 2013;28(5):783-92.
2. Liver Disease and Hepatic Encephalopathy, Scientific Product Monograph, 2012. Merz Pharmaceuticals.
3. Dbouk N, McGuire BM. Hepatic encephalopathy: a review of its pathophysiology and treatment. Curr Treat Options Gastroenterol. 2006;9(6):464-474.
4. Blanco Vela CI, Poo Ramirez JL. Efficacy of oral L-ornithine L aspartate in cirrhotic patients with hyperammonemic hepatic encephalopathy. Ann Hepatol 2011;10(2):55-59.